MEYLAN, France, April 8th, 2021 – KOELIS, an international leader in prostate fusion imaging, announces enrolling the first patients in the VIOLETTE clinical trial (NCT04582656) on 3D fusion imaging-guided focal ablation of prostate cancer.
Prostate cancer is the most common cancer among men. In the past ten years, prostate imaging developments allowed to reduce overdiagnosis and overtreatment of prostate cancer. Focal treatment is a novel approach aiming at treating only the prostatic lesion while sparing healthy tissue. KOELIS 3D technologies are fundamental assets to precisely deliver and execute focal treatment.
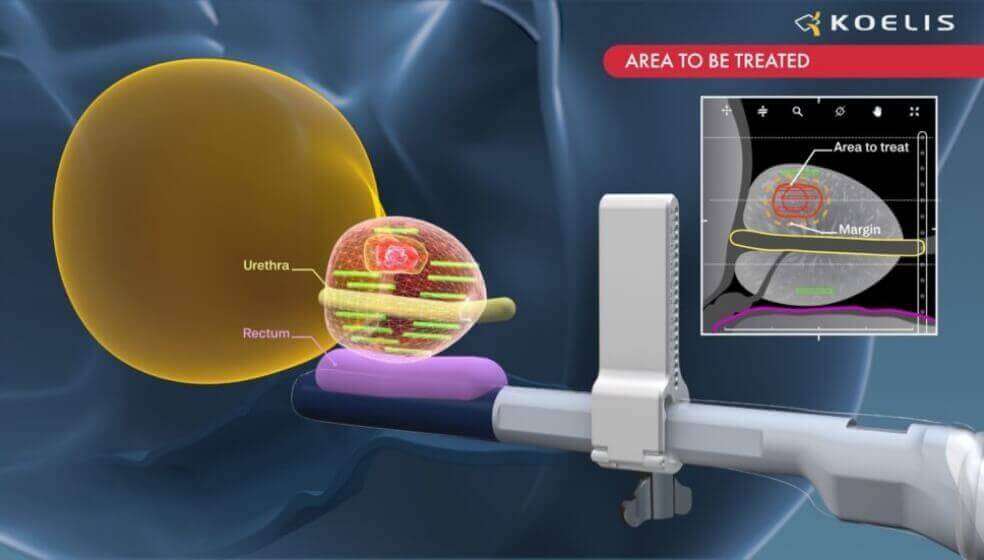
The VIOLETTE study aims at evaluating in selected patients the safety and efficacy of a novel approach in prostate focal therapy. In line with the 2019 Kyoto consensus, the novelty lies in the precise patient selection by multimodal imaging tools and 3D cartography technologies. Localizing and quantifying the “index lesion” in 3D help planning, guiding, and monitoring the local application of an ablative energy.
KOELIS is the inventor of the OBT Fusion®, a patented technology that provides a unique precision to guide diagnostic and therapeutic needles to identified areas of the prostate. It is integrated to KOELIS medical device Trinity®, a versatile fusion imaging platform, enabling the guidance of focal ablations of the prostate. Using this combination of unique technologies, focal ablation of the prostate with thin microwave needles can be performed with precision and confidence in less than one hour. All devices developed by KOELIS and used in the VIOLETTE study are already CE-marked.
VIOLETTE is a multicentric prospective non-comparative interventional trial that will enroll at least 65 patients from 8 European urological centers (6 in France) and store data into a dedicated registry. The primary outcome is the absence of cancer in the treated area 12 months after treatment. Importantly, secondary outcomes will be measured regarding patient Quality of Life and satisfaction. Multi-parametric MRI, digital pathology and 3D targeted biopsy will be used to monitor patients. The protocol was built in accordance with the latest international[1] and FDA guidances[2]. VIOLETTE is a phase 2 clinical study following the FOSTINE feasibility and precision trial led on 10 patients in Paris Cochin hospital from 2017 to 2019.
“We are very excited to lead a world premiere phase 2 study following the success of our pilot study ‘FOSTINE’ with results to be published this year. KOELIS has been visionary and instrumental in designing a new targeted focal approach as well as in gathering some of the most experienced experts in the field”, said Prof. Nicolas Barry Delongchamps, M.D., Ph.D., Professor of urology at Paris Cochin hospital and Principal Investigator of the VIOLETTE trial.
The first intervention was performed on March 9th at Clinique Nantes Atlantis, Saint-Herblain, France by Dr. Eric Potiron, who reports immediate physician and patient satisfaction.
“We realize that our efforts in developing technology and clinical evidence support the emergence of focal therapy as a paradigm shift in prostate cancer care globally, and that makes us very proud. The KOELIS teams in France, in the USA, and in Asia, with their distributors, are committed to diffusing our technology globally”, concluded Antoine Leroy, Ph.D., founder and CEO of KOELIS.
Intermediate results of the VIOLETTE study are expected in 2022 and final results in 2023. In the meantime, several monocentric and multicentric studies are in preparation in other countries.
About KOELIS:
KOELIS is the world leader in fusion imaging technology, with a focus on prostate interventions. KOELIS Trinity is a unique, integrated, versatile platform with 3D ultrasound capabilities and a multi-patented software fusion technology called Organ Based Tracking®. 3DUS and OBT Fusion® enable precise 3D prostate biopsy cartography, as well as other prostate interventions beyond biopsy. With Violette, KOELIS aims at offering a complete solution from diagnostics to focal treatment of Prostate Cancer. KOELIS is a privately owned Medtech company based in Grenoble, France, with a subsidiary in Princeton, USA, and an office in Hong Kong. In more than 30 countries around the world, KOELIS grows innovation in prostate care to improve physicians’ precision and patients’ quality of life.
[1] Lebastchi et al. – Eur Urol 2020 – Standardized Nomenclature and Surveillance Methodologies After Focal Therapy and Partial Gland Ablation for Localized Prostate Cancer: An International Multidisciplinary Consensus
[2] FDA guidances on Clinical Investigations for Prostate Tissue Ablation Devices: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/clinical-investigations-prostate-tissue-ablation-devices